Author: shiaulab
Our protocol was ranked a most-read this year in immunology!
Now reprinted in Immunology Selections as part of a curated series of 17 methods most used by Bio-protocol’s community over the past year.
Check it out here: https://en.bio-protocol.org/ebooks.aspx?source=site.
Our paper just published in new open access Nature portfolio journal Communications Biology!
Peripheral NOD-like receptor deficient inflammatory macrophages trigger neutrophil infiltration into the brain disrupting daytime locomotion
Victoria Kwon1,3, Peiwen Cai1,3, Cameron T. Dixon1,3, Victoria Hamlin1, Caroline G. Spencer1, Alison M. Rojas1, Matthew Hamilton 1 & Celia E. Shiau 1,2,3 ✉
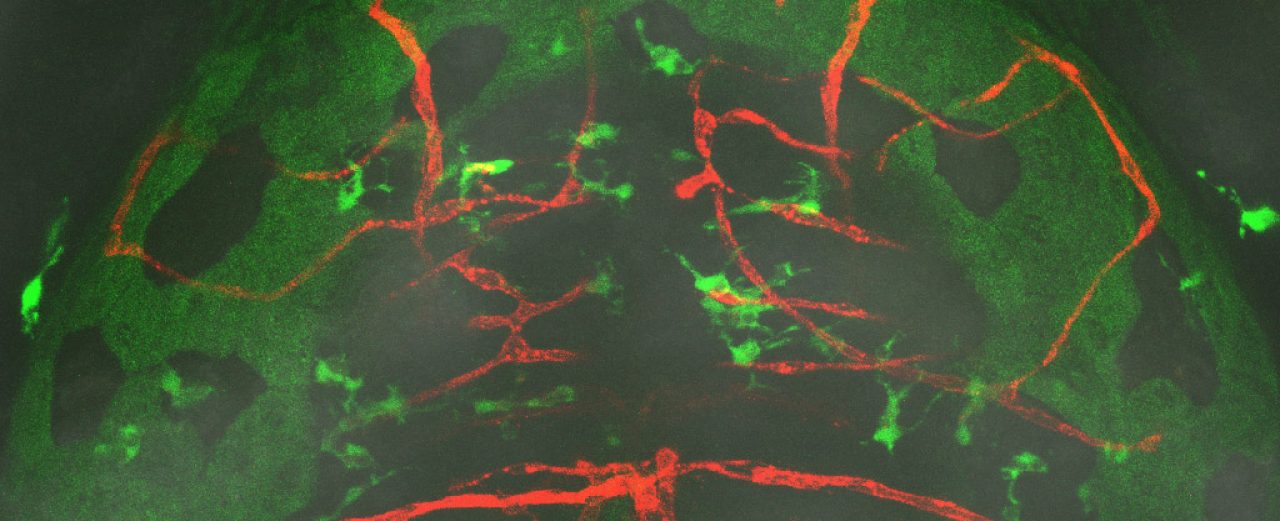
Inflammation is known to disrupt normal behavior, yet the underlying neuroimmune interactions remain elusive. Here, we investigated whether inappropriate macrophage-evoked inflammation alters CNS control of daily-life animal locomotion using a set of zebrafish mutants selected for specific macrophage dysfunction and microglia deficiency. Large-scale genetic and computational analyses revealed that NOD-like receptor nlrc3l mutants are capable of normal motility and visuomotor response, but preferentially swim less in the daytime, suggesting possible low motivation rather than physical impairment. Examining their brain activities and structures implicates impaired dopaminergic descending circuits, where neutrophils abnormally infiltrate. Furthermore, neutrophil depletion recovered daytime locomotion. Restoring wild-type macrophages reversed behavioral and neutrophil aberrations, while three other microglia-lacking mutants failed to phenocopy nlrc3l mutants. Overall, we reveal how peripheral inflammatory macrophages with elevated pro-inflammatory cues (including il1β, tnfα, cxcl8a) in the absence of microglia co-opt neutrophils to infiltrate the brain, thereby potentially enabling local circuitry modulation affecting daytime locomotion.
Sneak peek on a really cool macrophage-brain-behavior study out on preprint!
Our paper “Peripheral NOD-like receptor deficient inflammatory macrophages trigger neutrophil infiltration disrupting daytime locomotion” that reveals the impact of macrophage inflammation on spontaneous locomotor change is published early as a preprint on BioRxiv. Check it out here: https://doi.org/10.1101/2021.10.27.466033. This was made possible by a large team effort over a long timeline to accomplish such an immense study that spans multiple disciplines.
Check out our new paper at iScience!
Our study provides detailed characterizations of intestinal macrophages and demonstrate diverse populations in zebrafish for further investigations. Read here.

Check out our newest paper in eLife!
Check it out here: https://elifesciences.org/articles/58191
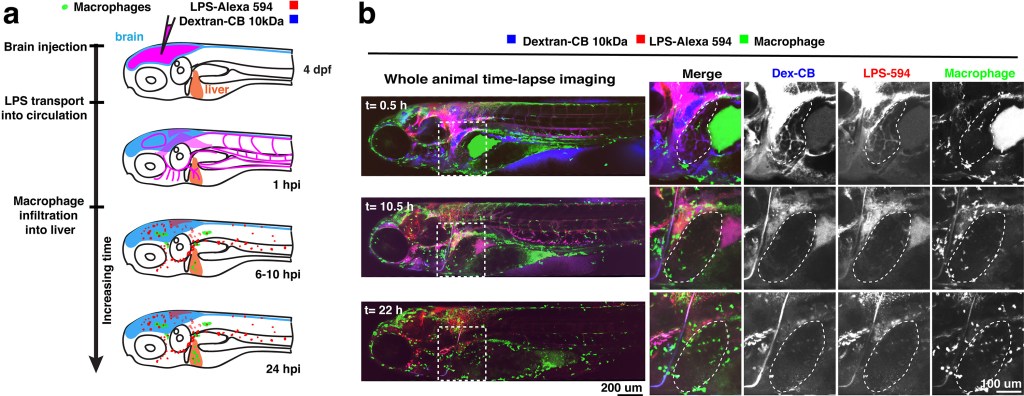
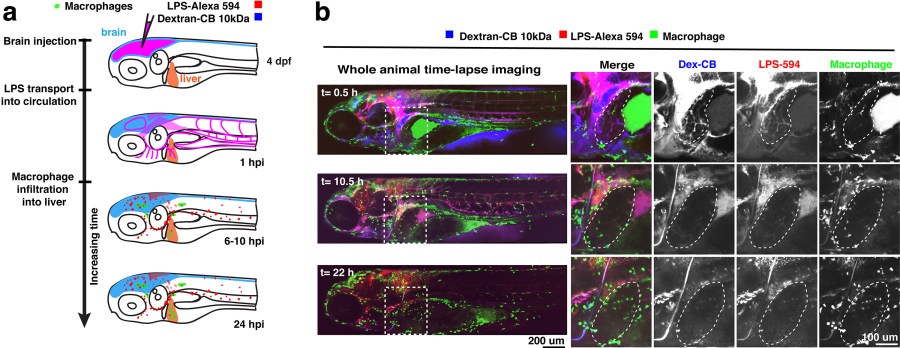
Microinjection of traceable LPS into the brain leads to drainage of LPS molecules into circulation, and causes active infiltration of peripheral macrophages into the liver. (a) Schematic. (b) Whole-body in vivo imaging analysis. Dotted line demarcates the liver.
Dr. Celia Shiau awarded 5-year NIH grant
Dr. Shiau awarded a 5-year Outstanding Early-Stage Investigator MIRA grant from NIGMS! The MIRA mechanism provides flexibility for NIGMS-supported investigators to pursue innovative and important research directions. The Shiau Lab will use this award to study the genetic and metabolic regulation of macrophage activation in vivo using zebrafish and other systems. This has implications for the design of macrophage-targeted technologies for controlling inflammatory processes and addressing health challenges related to immune dysregulation.